- Home
- Google Trends
- April 26, 2026
- , are pleased to announce that from 1 august 2024, onpattro® patisiran is listed on the pharmaceutical benefits scheme pbs for the treatment of hereditary transthyretinmediated amyloidosis hattr amyloidosis in adult patients, with stage 1 or stage 2 polyneuropathy.
, are pleased to announce that from 1 august 2024, onpattro® patisiran is listed on the pharmaceutical benefits scheme pbs for the treatment of hereditary transthyretinmediated amyloidosis hattr amyloidosis in adult patients, with stage 1 or stage 2 polyneuropathy.
Why is , are pleased to announce that from 1 august 2024, onpattro® patisiran is listed on the pharmaceutical benefits scheme pbs for the treatment of hereditary transthyretinmediated amyloidosis hattr amyloidosis in adult patients, with stage 1 or stage 2 polyneuropathy. trending?
, are pleased to announce that from 1 august 2024, onpattro® patisiran is listed on the pharmaceutical benefits scheme pbs for the treatment of hereditary transthyretinmediated amyloidosis hattr amyloidosis in adult patients, with stage 1 or stage 2 polyneuropathy. - More News on LatestLY

‘Hello Bachhon’ Review: Vineet Kumar Singh’s Alakh Pandey Netflix Biopic Series Is Predictable and Feels More Like an Ad Campaign, Say Critics

OTT Releases This Week: ‘Subedaar’, ‘Young Sherlock’, ‘Gandhi Talks’, ‘War Machine’ and More To Stream on Netflix, Prime Video, Apple TV+

Maharashtra: Government Officials Will Have To Say 'Vande Mataram' Instead of Hello For Work-Related Phone Calls, Says Minister Sudhir Mungantiwar (Watch Video)
(Know why , are pleased to announce that from 1 august 2024, onpattro® patisiran is listed on the pharmaceutical benefits scheme pbs for the treatment of hereditary transthyretinmediated amyloidosis hattr amyloidosis in adult patients, with stage 1 or stage 2 polyneuropathy. is trending in Google Trends today, on March, 08 2026. Check latest news and articles on , are pleased to announce that from 1 august 2024, onpattro® patisiran is listed on the pharmaceutical benefits scheme pbs for the treatment of hereditary transthyretinmediated amyloidosis hattr amyloidosis in adult patients, with stage 1 or stage 2 polyneuropathy. updated real-time on Google Trends and LatestLY)
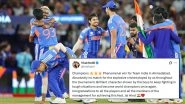
Crowd at Narendra Modi Stadium Sings 'Maa Tujhe Salaam' After India Win T20 World Cup 2026 Final, Video Goes Viral
Mallikarjun Kharge Slams PM Narendra Modi Over US Ties, Calls Donald Trump a ‘Dictator’ and Prime Minister His ‘Slave’
Player of the Tournament Sanju Samson Credits Sachin Tendulkar After his Success in ICC T20 World Cup 2026
Sanju Samson Wins Player of the Tournament Award in ICC T20 World Cup 2026
WhatsApp Deleted Messages: Know How to Restore Your Chat History on Android and iPhone
Apple MacBook Ultra, New Foldable iPhone and Smart AirPods Launching This Year: Report
Read More
Parliament Budget Session 2026: Shashi Tharoor Says No-Confidence Motion Against Lok Sabha Speaker Om Birla Will Let Opposition Present Concerns Despite Likely Defeat
US-Israel vs Iran War: Debris From Aerial Interception Falls on Vehicle in Dubai’s Al Barsha Area, Pakistani Driver Killed (Watch Video)
‘To Take a Little Pressure Off’: Donald Trump Says on US Granting ‘Permission’ to India to Accept Russian Oil
Gracie Mansion Bomb Attack: 2 Arrested After Ignited Devices Thrown Near Zohran Mamdani Residence, Conservative Activist Jake Lang Claims ‘Assassination Attempt’ During Anti-Muslim Protest (Watch Video)
US-Israel vs Iran: IRGC Retaliates, Strikes Haifa Refinery With Kheibarshekan Missiles After Attacks on Iranian Energy Infrastructure (Watch Videos)
Anirudh Ravichander REACTS to Marriage-Related Question Amid Kavya Maran Dating Rumours; His Response Goes Viral (Watch Video)
-
Did West Bengal Govt ‘Snub’ President Droupadi Murmu? Political Row Over Venue Change for Santal Conference
-
Did Barron Trump Buy USD 50 Million Worth of Oil Sector Stocks Before US and Israel Struck Iran? Know Truth Behind the Viral Social Media Rumours
-
MH370 Mystery: Ocean Infinity’s Search for Missing Malaysia Airlines Flight Concludes Without Findings; Families Urge Extension
-
IAF Pilot Anuj Vashisht Funeral: Fiancee and Fellow Pilot Bids Teary-Eyed Farewell to Squadron Leader Killed in Su-30 MKI Crash, Video Goes Viral












 Quickly
Quickly






